AQA / A Level
AQA A Level Chemistry Paper 2 2025 Mark Scheme Pdf
Download the official AQA A-Level Chemistry Paper 2 mark scheme for the June 2025 exam (7405/2). This is the final examiner-approved marking guide used during standardisation, containing full answers, detailed mark allocations, and examiner guidance on how marks are awarded.
The mark scheme provides complete solutions for all organic chemistry, analysis, and practical questions. For example, **Question 1 (page 11–14)** confirms the mechanism as *free-radical substitution* with UV light, followed by detailed propagation steps involving chlorine radicals and intermediate species.
Organic mechanisms are fully explained. **Question 2 (page 15–16)** includes stereoisomerism answers such as *Z-3-methylpent-2-ene* and explains restricted rotation around the C=C bond. It also shows the correct electrophilic addition mechanism with formation of a tertiary carbocation and nucleophilic attack by Br⁻.
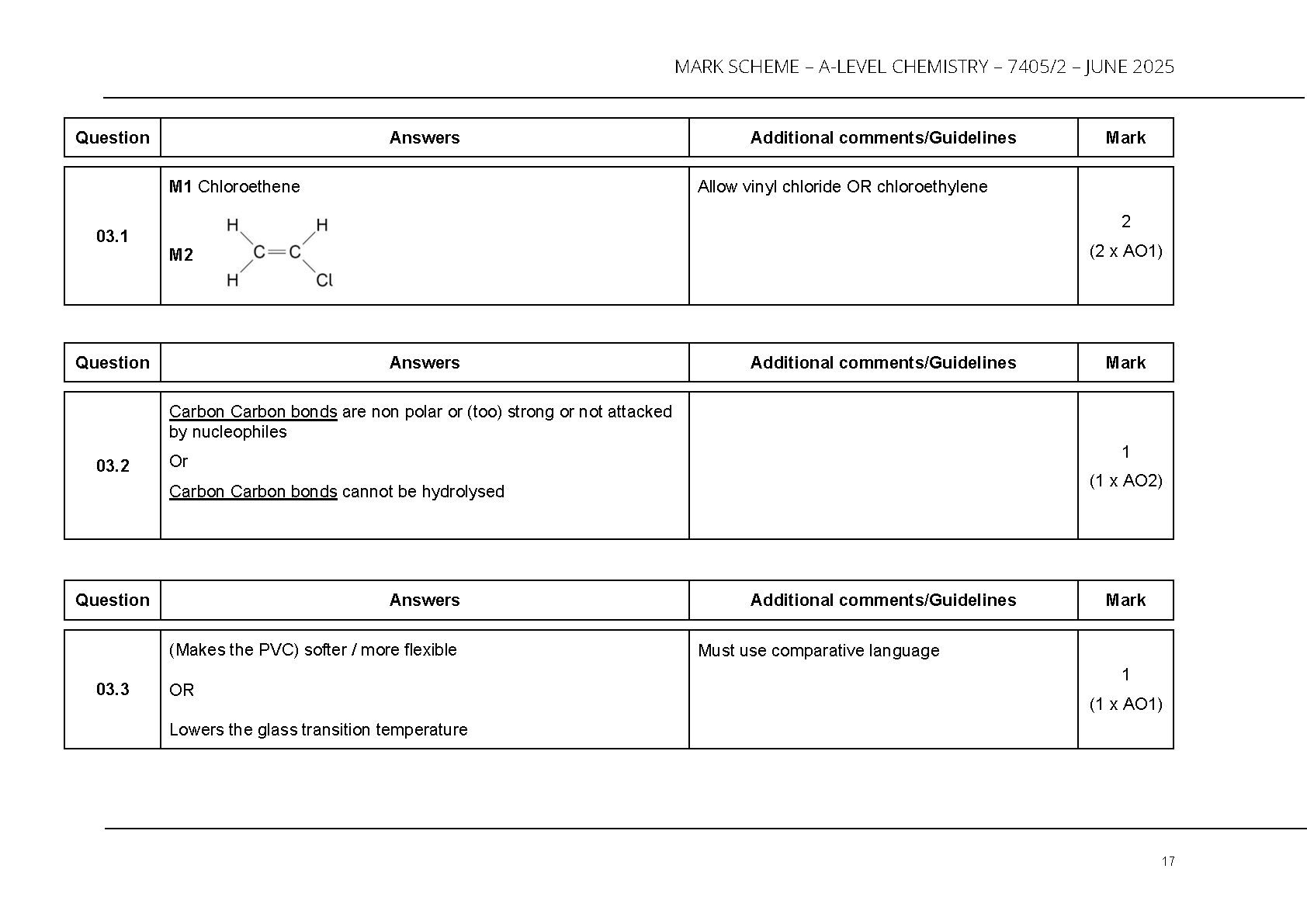
Polymer chemistry answers are clearly structured. **Question 3 (pages 17–21)** includes:
- Identification of chloroethene as the PVC monomer (page 17)
- Explanation of PVC’s non-biodegradability due to strong C–C bonds
- Plasticiser effects (increased flexibility)
- Mechanisms such as nucleophilic addition–elimination (page 18)
- Detailed curly-arrow mechanisms for nylon formation (pages 20–21)
The mark scheme also explains data interpretation. For example, **Table-based analysis in Question 3.6 (page 19)** shows that higher fat content leads to greater DEHA absorption due to non-polar interactions and van der Waals forces.
Practical chemistry questions are fully broken down. **Question 4 (pages 22–24)** includes:
- Safety reasoning (toxic gases like N₂O)
- Role of nitric acid as an oxidising agent
- Full recrystallisation method steps (dissolve hot, filter, cool, recrystallise, dry)
- Experimental errors in melting point apparatus (Figure 4, page 12 of paper)
Aromatic chemistry is covered in depth. **Question 5 (pages 25–26)** includes:
- Balanced nitration equation for TNT formation
- Full calculation yielding **955 g of TNT**
- Mechanism showing formation of the nitronium ion (NO₂⁺) and electrophilic substitution
Advanced spectroscopy and analysis are also included:
- **Question 6 (page 27):** NMR solvent reasoning (D₂O vs CCl₄), amino acid structure, and protein secondary structures (α-helix → β-pleated sheet)
- **Question 7 (pages 28–30):** Molecular formula determination (C₆H₁₂) and IR/NMR interpretation, including identification of C=O groups and splitting patterns
- **Question 8 (page 31–32):** Gas calculations using PV = nRT with final Mr = **135**, plus uncertainty analysis
- **Question 9 (pages 34–36):** Rates, experimental design, and activation energy calculation (~88 kJ mol⁻¹)
The mark scheme also includes detailed examiner guidance such as:
- Error carried forward (ECF) rules
- “Best fit” marking for extended responses
- Level-of-response criteria for 7-mark spectroscopy questions (page 29)
- Strict requirements for organic structures, mechanisms, and nomenclature
This resource is essential for understanding how to gain full marks, master organic mechanisms, and apply spectroscopy and calculations accurately in A-Level Chemistry exams. Fully updated for the 2025/2026 academic year and optimised for instant download on markscheme.net.
This mark scheme is essential for Year 13 students preparing for the 2026 AQA A-Level Chemistry exams, particularly those focusing on Paper 2 (organic chemistry and analysis).
Teachers and tutors use this resource to standardise marking, provide accurate feedback, and model high-quality exam answers. It is also ideal for independent learners and retake students who want detailed step-by-step solutions and insight into examiner expectations.
The mark scheme provides complete solutions for all organic chemistry, analysis, and practical questions. For example, **Question 1 (page 11–14)** confirms the mechanism as *free-radical substitution* with UV light, followed by detailed propagation steps involving chlorine radicals and intermediate species.
Organic mechanisms are fully explained. **Question 2 (page 15–16)** includes stereoisomerism answers such as *Z-3-methylpent-2-ene* and explains restricted rotation around the C=C bond. It also shows the correct electrophilic addition mechanism with formation of a tertiary carbocation and nucleophilic attack by Br⁻.
Polymer chemistry answers are clearly structured. **Question 3 (pages 17–21)** includes:
- Identification of chloroethene as the PVC monomer (page 17)
- Explanation of PVC’s non-biodegradability due to strong C–C bonds
- Plasticiser effects (increased flexibility)
- Mechanisms such as nucleophilic addition–elimination (page 18)
- Detailed curly-arrow mechanisms for nylon formation (pages 20–21)
The mark scheme also explains data interpretation. For example, **Table-based analysis in Question 3.6 (page 19)** shows that higher fat content leads to greater DEHA absorption due to non-polar interactions and van der Waals forces.
Practical chemistry questions are fully broken down. **Question 4 (pages 22–24)** includes:
- Safety reasoning (toxic gases like N₂O)
- Role of nitric acid as an oxidising agent
- Full recrystallisation method steps (dissolve hot, filter, cool, recrystallise, dry)
- Experimental errors in melting point apparatus (Figure 4, page 12 of paper)
Aromatic chemistry is covered in depth. **Question 5 (pages 25–26)** includes:
- Balanced nitration equation for TNT formation
- Full calculation yielding **955 g of TNT**
- Mechanism showing formation of the nitronium ion (NO₂⁺) and electrophilic substitution
Advanced spectroscopy and analysis are also included:
- **Question 6 (page 27):** NMR solvent reasoning (D₂O vs CCl₄), amino acid structure, and protein secondary structures (α-helix → β-pleated sheet)
- **Question 7 (pages 28–30):** Molecular formula determination (C₆H₁₂) and IR/NMR interpretation, including identification of C=O groups and splitting patterns
- **Question 8 (page 31–32):** Gas calculations using PV = nRT with final Mr = **135**, plus uncertainty analysis
- **Question 9 (pages 34–36):** Rates, experimental design, and activation energy calculation (~88 kJ mol⁻¹)
The mark scheme also includes detailed examiner guidance such as:
- Error carried forward (ECF) rules
- “Best fit” marking for extended responses
- Level-of-response criteria for 7-mark spectroscopy questions (page 29)
- Strict requirements for organic structures, mechanisms, and nomenclature
This resource is essential for understanding how to gain full marks, master organic mechanisms, and apply spectroscopy and calculations accurately in A-Level Chemistry exams. Fully updated for the 2025/2026 academic year and optimised for instant download on markscheme.net.

Download AQA A Level Chemistry Paper 3 (7405/3) June 2025 Mark Scheme Pdf With Full A...

Download AQA A Level Chemistry Paper 3 (7405/3) June 2025 Question Paper Pdf. Free In...
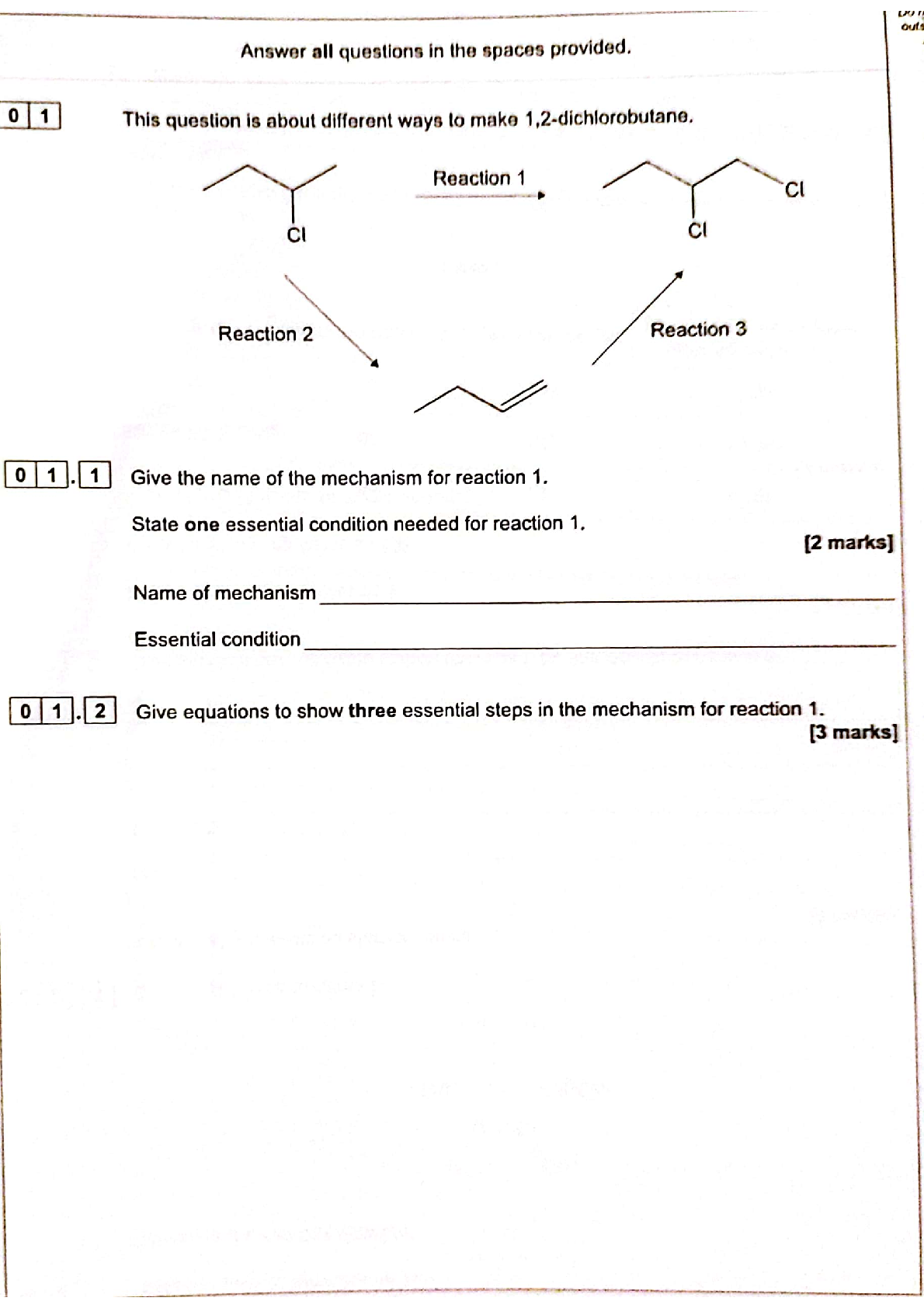
Download AQA A Level Chemistry Paper 2 (7405/2) June 2025 Question Paper Pdf. Free In...

Download AQA A Level Chemistry Paper 1 (7405/1) June 2025 Question Paper Pdf. Free In...