AQA / AS Level
AQA As Chemistry Paper 2 2025 Mark Scheme Pdf
Download the official AQA AS Chemistry Paper 2 mark scheme for the June 2025 exam (7404/2). This **final version (v1.0)** contains full worked answers, mark allocations, and detailed examiner guidance used during standardisation.
The mark scheme provides complete solutions for **Organic and Physical Chemistry**, including mechanisms, calculations, and multiple-choice answers:
**Section A (structured questions):**
- **Question 1 (pages 11–14): Halogenoalkanes**
- δ⁺ carbon attracts nucleophiles; lone pair attack mechanism
- SN1/SN2 mechanisms with correct curly arrows and intermediates
- Elimination mechanism forming alkenes (E/Z but-2-ene)
- Carbocation bond angle = **120°**
- Co-ordinate bond: both electrons donated from CN⁻ to C⁺
- **Question 2 (pages 15–16): Kinetics & bonding**
- Catalyst provides **alternative pathway with lower activation energy**
- Increased temperature → more particles exceed activation energy
- Hydrogen bonding: lone pairs on O, δ⁺/δ⁻ charges, dotted bond
- **Question 3 (page 17): Intermolecular forces (6-mark)**
- Tetrahedral shape: 4 bonding pairs repel equally
- CCl₄ non-polar (symmetrical), CHCl₃ polar
- Stronger London forces in CCl₄ due to more electrons → higher boiling point
- **Question 4 (pages 18–19): Organic structures & IR**
- Accepts multiple valid isomers of C₄H₆O₂
- IR interpretation: OH + C=C functional groups
- Polymer repeating unit of vinyl acetate
- **Question 5 (page 20): Mass spectrometry & gas calculations**
- High-resolution MS distinguishes molecules with different exact masses
- Ideal gas calculation:
- n ≈ **0.00895 mol**
- mass = **0.761 g**
- Mr = **85.0**
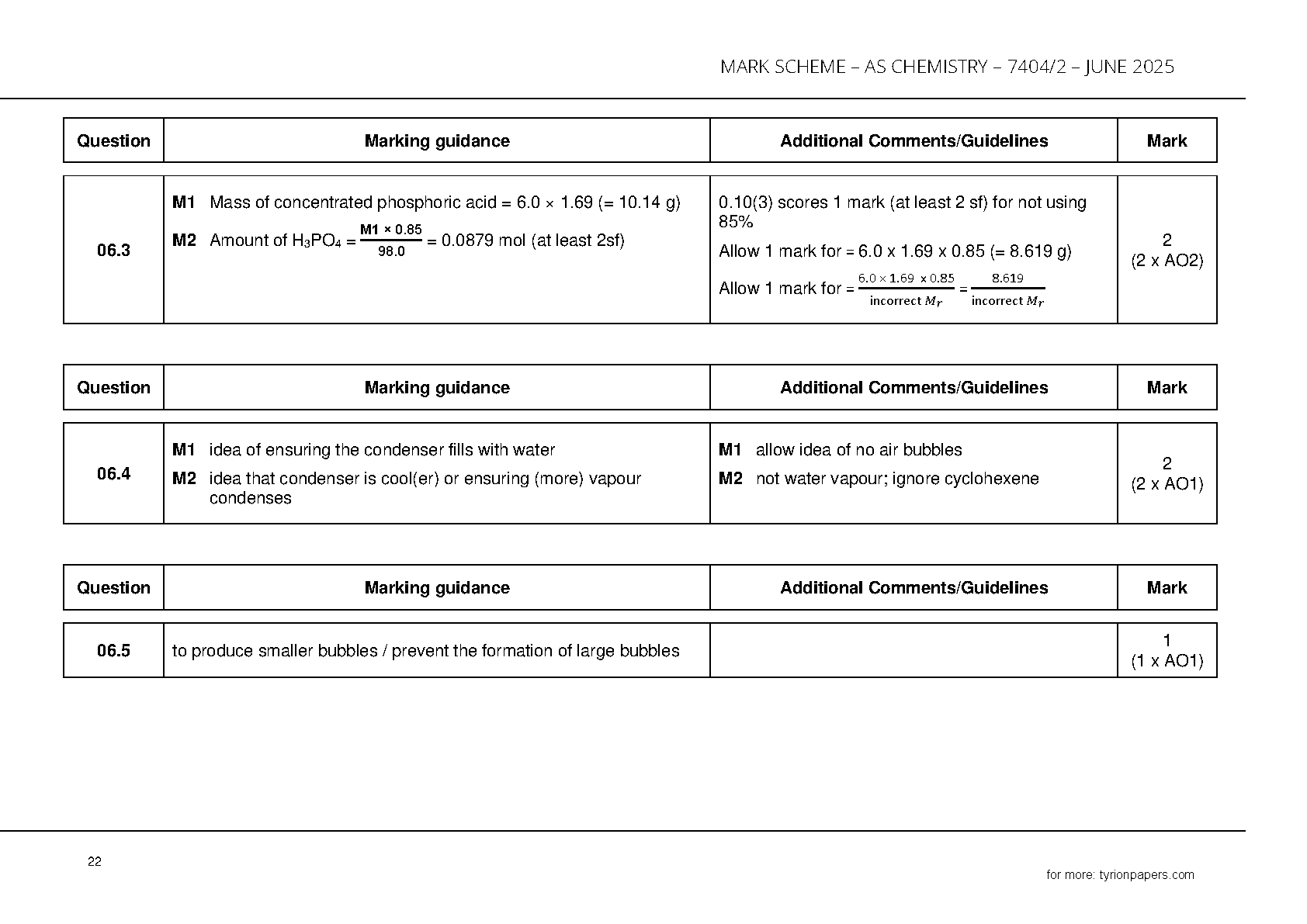
- **Question 6 (pages 21–24): Organic synthesis & practicals**
- Hydration mechanism (curly arrows: C=C → H⁺, nucleophilic attack, deprotonation)
- Amount of H₃PO₄ = **0.0879 mol** (from 6.0 cm³ sample)
- Condenser water enters at bottom → ensures full cooling
- Anti-bumping granules prevent large bubbles
- Electric heater used because mixture is **flammable**
- Closed system risk → pressure build-up; solution = vent
- Dehydration mechanism = **elimination**
- Combustion equation:
- C₆H₁₁OH + 8½O₂ → 6CO₂ + 6H₂O
- Carbon-neutral = no net CO₂ emissions
- **Question 7 (pages 25–26): Energetics**
- Enthalpy change = heat change at constant pressure
- q = mcΔT = **10621 J**
- Mass of ethanol = **0.356 g**
- Bond enthalpy expressions:
- Products: **2536n + 926**
- Alkane identified as **C₅H₁₂**
**Section B (multiple-choice answers, pages 27–28):**
All answers provided, for example:
- Q8: **B** (3-methylbutanal)
- Q10: **C** (C–C bonds broken in cracking)
- Q14: **D** (SO₂ has polar bonds)
- Q18: **A** (+51 kJ mol⁻¹)
- Q20: **C** (rate experiment conditions)
- Q22: **B** (enthalpy of formation equation)
The **marking guidance pages (2–10)** also explain:
- Error carried forward (ECF) rules
- “Right + wrong = wrong” list marking principle
- Organic structure and mechanism marking standards
- Level-of-response marking for extended questions
For example, the **table on page 4** shows how incorrect extra answers cancel correct ones in list questions, while pages 6–8 define strict rules for drawing organic structures and mechanisms.
This mark scheme is essential for understanding exactly how marks are awarded, improving exam technique, and mastering calculations, mechanisms, and extended responses for top grades.
Fully updated for the 2025/2026 academic year and optimised for instant download on markscheme.net.
This mark scheme is ideal for Year 12 students preparing for AQA AS Chemistry exams and Year 13 students strengthening organic and physical chemistry fundamentals.
Teachers and tutors use this resource to standardise marking, provide accurate feedback, and model full-mark answers. It is also perfect for independent learners aiming to understand examiner expectations in detail.

Download AQA As Chemistry Paper 2 (7404/2) May 2025 Question Paper Pdf. Free Instant...

Download AQA As Chemistry Paper 1 (7404/1) June 2025 Mark Scheme Pdf With Full Worked...

Download AQA As Chemistry Paper 1 (7404/1) May 2025 Question Paper Pdf. Free Instant...